Patients usually are not aware of it, but drive systems are always at their side: in prophylaxis when dentist’s use handtools with ultra-low vibrations, in diagnostics systems where medical imaging provides ultra-sharp images, in robot assisted incisions supporting surgeons, personally in rehabilitation devices or prosthetics. The range of these and other medical applications where failure must absolutely not happen is large. Whatever your medical application needs may be, our broad portfolio of drive system and accessories is always the right prescription.
For example, hand-held devices like in endodontics or surgical hand tools benefit from our highly efficient drives, optimized for high speed operations at up to 100.000 rpm whilst their heating-up is very slow, allowing a hand-tool that always remains in a comfortable temperature range. For those applications, where installation space is extremely tight, our high-torque drives with zero-backlash gearheads are as short and as light-weight as possible. And if your application needs to be autoclavable, we got that covered as well.
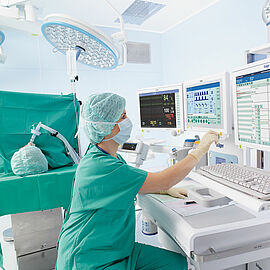
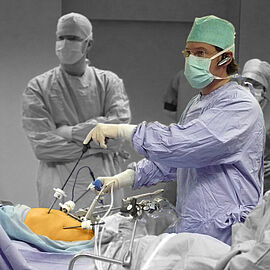
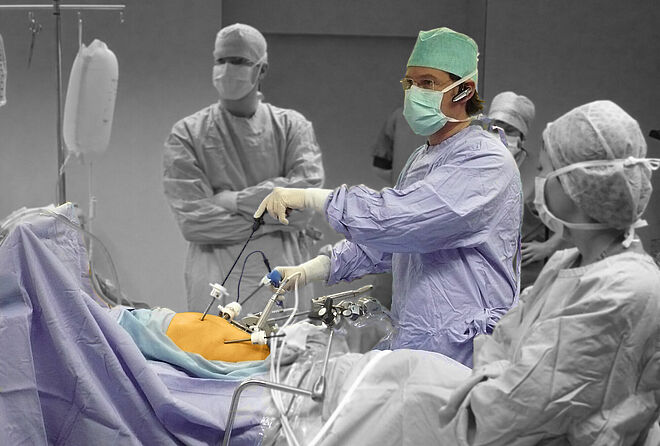
In the operating room, making the perfect cut is crucial for the success of the surgical procedure. To achieve that, surgeons can not only choose from surgical hand tools, but also from a wide range of surgical robotics. Their haptic feedback enables the operator to position the instruments very precise making the perfect cut. Thanks to ironless winding technology and flat speed-torque characteristics, our drive systems have all the necessary properties for surgical robotics. Powerful motor families, supplemented by the extensive range of gears, optical, magnetic or absolute encoders as well as speed and motion controllers, are ideal for demanding robotic applications not only in medicine but also in many other areas.
FAULHABER drive systems offer further benefits, for example our quiet drives allow users of prosthetics to master their busy daily life without having to worry about battery life or nuisances from noise, on the following pages, we will show you how our drives support your medical application as well.